IBM announced a scientific achievement involving the creation of a molecule with a half-Möbius topology, a process aided by an algorithm partially run on a quantum computer. The research highlights progress in synthetic chemistry using modern computational tools and the gradual advancement of quantum computing toward practical applications.
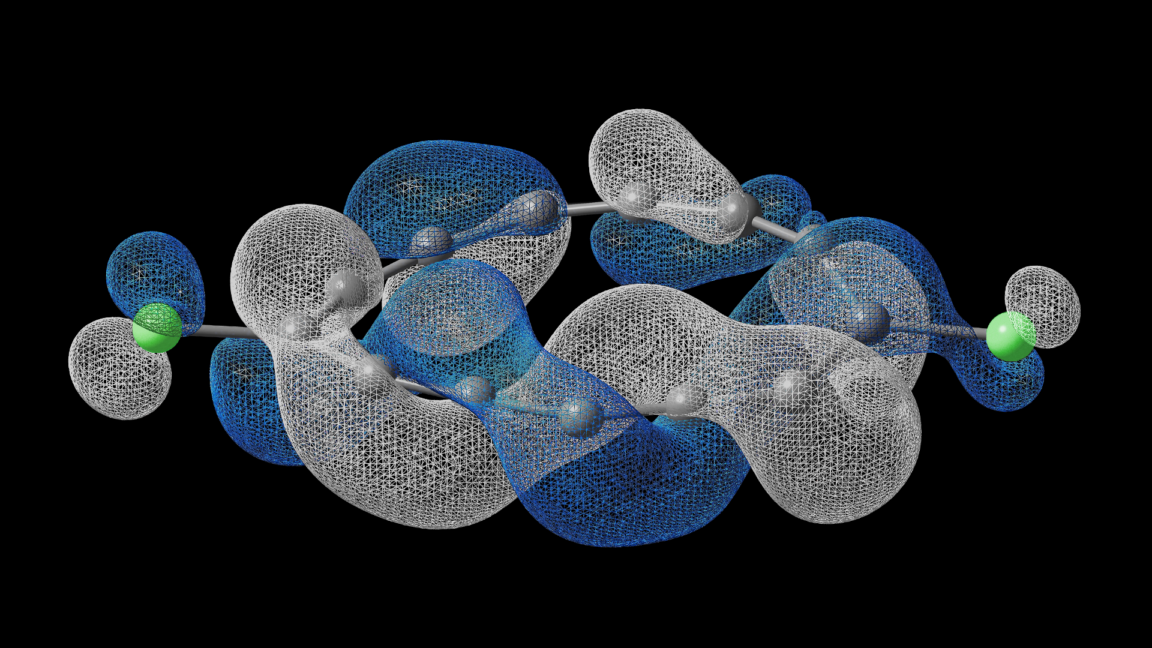
The paper delves into complex chemistry, using benzene's electron orbitals as a starting point to explain how molecular structures can be manipulated. By altering orbital arrangements, scientists can create twisted electron pathways, similar to a Möbius strip, where an electron must travel two loops around the molecule to return to its starting point.
The main topics covered are a novel chemical synthesis, the role of quantum computing in research, and the principles of electron orbital topology in molecules.
Last week, IBM trumpeted its contributions to a rather unusual paper: the production of a molecule with a half-Möbius topology, assisted by an algorithm run in part on a quantum computer. There was, to put it mildly, a lot going on in this paper, and it took a little while to digest. But it’s interesting in what it says about the sorts of chemistry that we can construct with tools developed over the past several decades, as well as how quantum computation is inching toward utility.
But getting the full picture requires about three different stories, so we’ll go through each of them separately before bringing the big picture together.
Orbitals with a twist
Those of you who can still dredge up your high school chemistry lessons probably remember benzene, a six-carbon ring with alternating single and double bonds that kept all the carbons locked into a single plane, creating a flat molecule. What you are a bit less likely to remember is that the double bonding is mediated by orbitals that extend vertically above and below the nucleus of the carbon atoms. Thanks to the alternating single-double nature of the bonds, electrons in these orbitals end up delocalized; the differences between the bonds become a bit irrelevant, and the molecule is best viewed as having some of its electrons floating around in a cloud. The same would hold true for even larger molecules with the same sort of bonding arrangement.
The way the orbitals are arranged in benzene, any electrons that circulate around the molecule will end up back where they started. By linking other atoms to the carbon ring, it’s possible to place orbitals at different angles from the vertical. With the right combination of additional orbitals, it’s possible to twist things so that an electron starts out on top of the molecule (assuming the ring is laid flat on a surface) but ends up at the bottom by the time it completes a full circuit around the ring. It would have to do a second loop around the ring to end up back where it started.